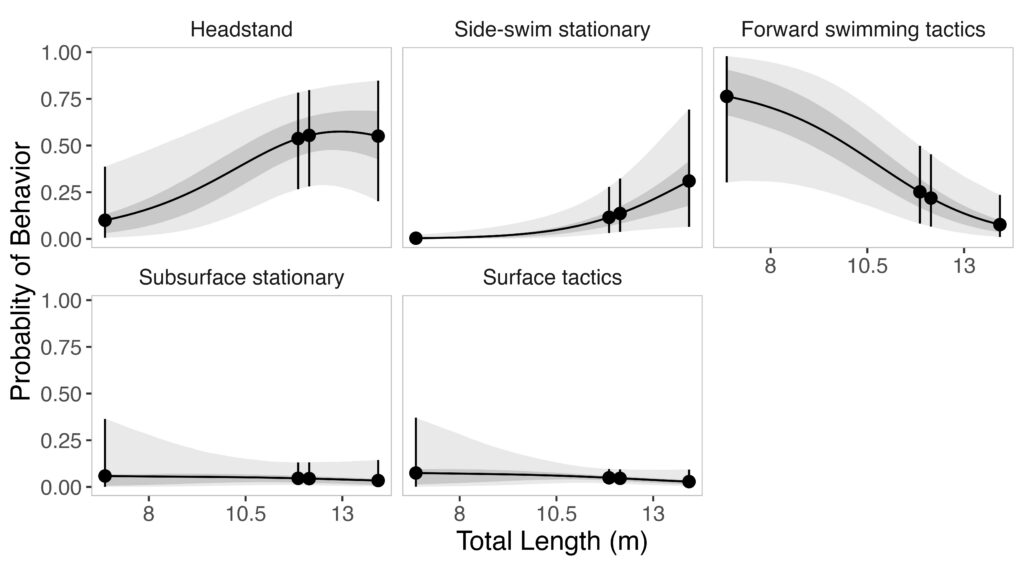
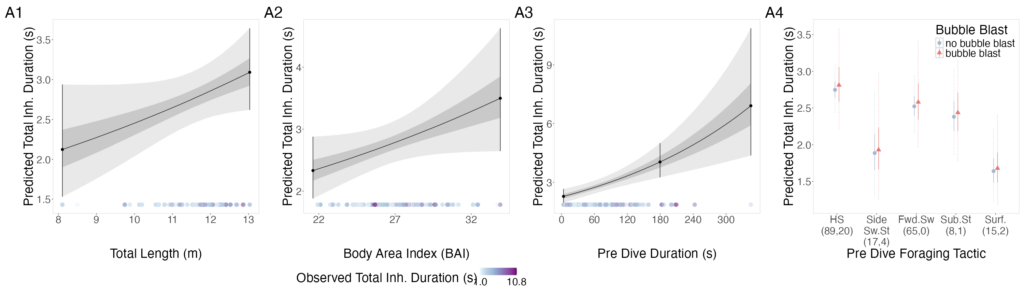
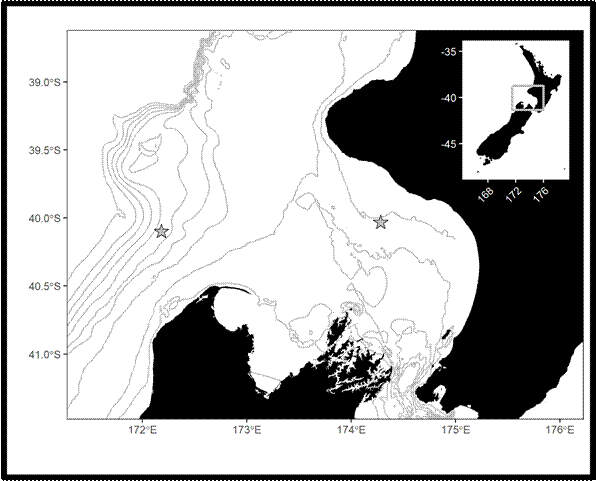
Mariam Alsaid, GEMM Lab Research Technician
It’s been three years since I last wrote a blog post for the GEMM Lab. At the time, I was wrapping up a wonderful summer filled with science, sun, and new friends and was eager to share all that I had learned and accomplished during my 10 weeks working on the Oregon coast as part of Hatfield Marine Science Center’s Research Experience for Undergraduates (REU). My love and concern for our oceans and its animals has always been the driving force of my academic pursuits. I am so thankful to this REU – funded by the US National Science Foundation – for introducing me to marine science research as a career path.
Alongside the mentorship of Drs. Dawn Barlow and Leigh Torres, I investigated the occurrence of sei whales, an endangered species of baleen whale, offshore of Oregon. The sei whale’s global population is believed to have dropped by 80% due to whaling in the 19th and 20th centuries and little is known about their contemporary population status, especially in the Northeast Pacific (Horwood, 2018; Nieukirk et al., 2020). Sei whales have occasionally been observed by the GEMM Lab on research cruises and we were curious about when and how often they were in our neck of the woods. Despite being massive animals (the 3rd largest rorqual in the world!), these animals are challenging to study due to their low population densities and visual similarities to fin whales (Horwood, 2018, Reeves et al., 1998). Luckily, sei whales produce characteristic vocalizations that can help distinguish them from other baleen whales that call at similar frequency bandwidths (e.g. blue, fin, and humpback whales).

This is where bioacoustics – the study of sounds produced by living organisms – comes in. We are able to study this elusive species by deploying hydrophones (underwater microphones) offshore of Oregon and reviewing recorded data in search of sei whale calls. (See this blog post if you’re curious about our methods and findings!).
Since 2021, the GEMM Lab and the K. Lisa Yang Center for Conservation Bioacoustics at Cornell University have collaborated to deploy three hydrophones for continuous acoustic recording along the Newport Hydrographic Line as part of the Holistic Assessment of Living marine resources off Oregon (HALO) project. These hydrophones are bottom mounted to the seafloor and are deployed 20, 45, and 65 nautical miles offshore of Newport, OR. HALO cruises are scheduled to refurbish and redeploy these instruments every six months or so. These cruises are also an opportunity to conduct visual surveys of cetaceans and seabirds in between deployments.

I immediately fell in love with bioacoustics and was enraptured by the diversity of clicks, pulses, and moans made by marine animals that flood our oceans. Hydrophones are able to record sounds in locations and frequency ranges that we would never naturally get to experience. Baleen whales are especially fascinating because they tend to produce infrasonic calls (i.e., <20 Hz) to communicate over large distances. These calls occur at frequencies below our hearing range and are only audible when sped up. Listening to these recordings feels like a window into a secret world that few have ventured.
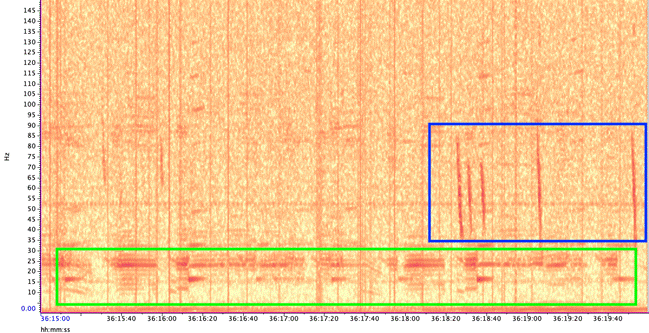
Figure 3. Period of high vocal activity from blue whales (A, B, and D calls), fin whales (20 Hz pulses) , and rockfish (pulses). Audio was sped up by 5x.
After completing my undergraduate degree, I participated in a bioacoustics research fellowship hosted by the US Forest Service and Oak Ridge Institute for Science and Education. During this time, I explored the world of terrestrial acoustics by leading a research project on American pika occupancy in the Oregon and Washington Cascades. Forest soundscapes have a different beauty to them. While periods of quiet are rare in the ocean, this is even scarcer on the surface. Bird songs, insect buzzing, human chatter, rainfall, and highway noise constantly fill our soundscapes, sometimes simultaneously. This experience processing terrestrial sound gave me a newfound appreciation for the world around me and the sounds I’ve become so accustomed to hearing. Nevertheless, I found myself yearning for the ocean and its secrets.
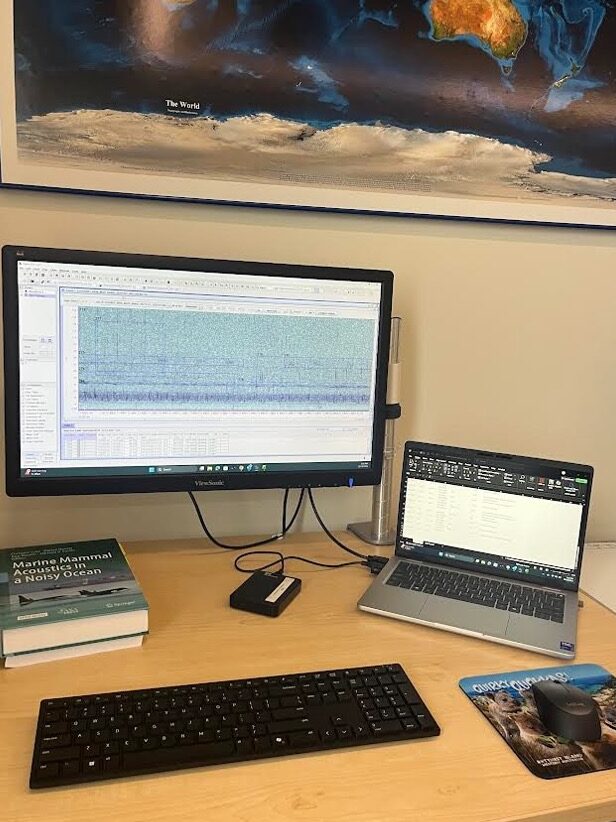
I was invited back to the Marine Mammal Institute last fall as the GEMM Lab’s baleen whale acoustic analyst to continue working on the HALO project. I dove back into the data collected from our hydrophones, this time targeting blue, fin, and humpback whale calls in addition to sei whales. My efforts were also broadened to all five years of recording data instead of just one. Because manually reviewing large amounts of acoustic data can be cumbersome, I trained artificial intelligence classifiers using the BirdNET convolutional neural network to make acoustic data processing more efficient. After much iterative trial and error, I was able to develop a set of four classifiers, each specialized to detect one of our four target species. The first classifier was trained to target blue whale A, B, and D calls; the second targets fin whale 20 Hz and 40 Hz pulses; the third targets sei whale downsweeps; and the fourth targets humpback song and single non-song grunts. Once I evaluated that the classifiers were performing with high precision and recall rates, I used them to predict when calls occurred in the five years of recording data. I am currently in the process of validating the hundreds of thousands of predictions to generate detection histories of these species of baleen whales in this region. This information will be incorporated with various oceanographic datasets to get a better understanding of baleen whale distribution patterns and habitat preferences in the Northeast Pacific.
I will be wrapping up my contract with the Marine Mammal Institute next month and I am endlessly grateful to the GEMM Lab for shaping me into the marine mammalogist I am today. I will look back on my time here fondly as I begin this next chapter of my life as a PhD student at the University of Washington’s School of Oceanography this fall. I am excited to share that I will continue to use acoustics to study baleen whales in the Northeast Pacific during my PhD! Although I dread the thought of having to say goodbye to Oregon and the Hatfield community, I thankfully won’t have to say goodbye to the whales.
Works cited
Horwood, J. (2018). Sei Whale – Balaenoptera borealis. Encyclopedia of Marine Mammals
(Third Edition), 845-847. https://doi.org/10.1016/B978-0-12-804327-1.00224-7
Nieukirk, S. L., Mellinger, D. K., Dziak, R. P., Matsumoto, H., & Klinck, H. (2020). Multi-year
occurrence of sei whale calls in North Atlantic polar waters. The Journal of the
Acoustical Society of America, 147(3), 1842-1850. https://doi.org/10.1121/10.0000931
Reeves, R. R., Silber, G. K., Payne, P. M. (1998). Draft recovery plan for the fin whale
Balaenoptera physalus and sei whale Balaenoptera borealis. Office of Protected
Resources, National Marine Fisheries Service, National Oceanic and Atmospheric
Administration.