Updated spore counts are available for central Oregon, the Lower Columbia Basin, and the Grande Ronde Valley of Oregon:




Updated spore counts are available for central Oregon, the Lower Columbia Basin, and the Grande Ronde Valley of Oregon:



Honeydew has been observed in non-treated, artificially-infested plots at the Hermiston Agricultural Research and Extension Center.
In contrast to airborne ascospores, honeydew is contact-, splash- or insect-dispersed.
The presence of honeydew at harvest can make swathing and combining more difficult. Late season scouting and field monitoring will help to develop harvesting/seed cleaning schedules and identify potential problem fields or areas that will need increased monitoring in the 2021 season.
The easiest way to scout for honeydew is to gently brush the flowers and developing seeds with your hand as you walk the field – if you feel a sticky substance, it may be honeydew from ergot. Aphids, if present, can also produce a sticky substance called honeydew, so, be careful not to confuse ergot honeydew with honeydew secreted by these insect pests.
Updated spore counts are now available for the lower Columbia Basin of Washington. Compared to other locations we are seeing low, but steady counts in the area:

Spore production appears to be ramping up in the Lower Columbia Basin of Oregon:


This research is funded by the Washington Turfgrass Seed Commission, the Oregon Seed Council, the Eastern Oregon Kentucky Bluegrass Working Group, and the Oregon Department of Agriculture Alternatives for Field Burning Research Financial Assistance Program.
Updated spore counts are available for the central Oregon area:

This research is funded by the Washington Turfgrass Seed Commission, the Oregon Seed Council, the Eastern Oregon Kentucky Bluegrass Working Group, and the Oregon Department of Agriculture Alternatives for Field Burning Research Financial Assistance Program.
Updated spore counts are available for the Grande Ronde Valley. Large numbers of spores were intermittently detected at the site:

This research is funded by the Washington Turfgrass Seed Commission, the Oregon Seed Council, the Eastern Oregon Kentucky Bluegrass Working Group, and the Oregon Department of Agriculture Alternatives for Field Burning Research Financial Assistance Program.
Updated spore counts are available for the central Oregon area:

Spore traps have detected the presence of airborne ergot ascospores around Kentucky bluegrass production areas in the Grande Ronde Valley.
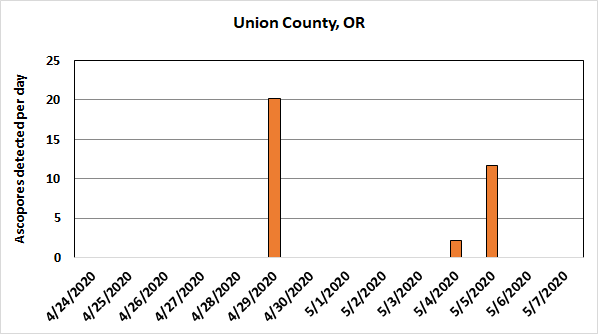
Spore counts are relatively low and sporadic compared to other sites. However, a relatively few number of ascospores can begin an infection, and the honeydew (secondary) stage of the disease can amplify the disease in a field before harvest. Our research has shown that, at least in some cases, up to 30% of infections can be caused by honeydew.
It is also important to note that the spore traps currently being used at all sites sample a relatively small volume of air (about 3,800 gallons of air/day). Consequently, the ascospores that are detected and reported likely represent a small proportion of the total number in the area.
Spore trap results confirmed the presence of airborne ergot ascospores in the Lower Columbia Basin of Oregon (top) and Washington (bottom):


The spore counts presented above are intended to show daily trends in spore production in their respective areas and do not necessarily indicate inoculum pressure in your field(s). Spore production can vary from field to field, and inoculum pressure tends to be higher in older fields with a history of ergot in the previous season, or in new fields planted next to established fields with a history of ergot.
A predictive model for ergot ascospores was developed for the Lower Columbia Basin of Oregon that uses accumulated degree-days (beginning January 1, with a base temperature of 50°F and upper threshold temperature of 77°F) to forecast when ascospores are likely to be present.
Accumulated degree-days as of May 10 were 435 in Hermiston, OR (orange line). According to the model, most ascospores are produced in the Lower Columbia Basin when accumulated degree-days are between 414 and 727.
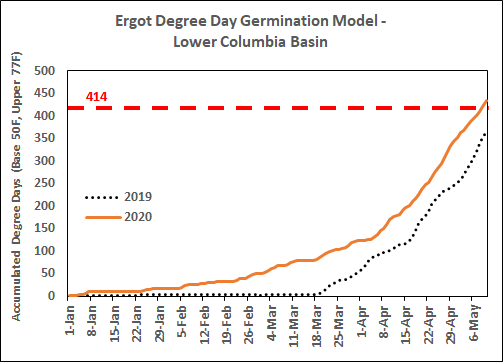
At this time in 2019, accumulated degree-days were 365 (dotted line).