Dr. Patty Skinkis, Viticulture Extension Specialist & Professor, Dept. of Horticulture, OSU
Our OSU Viticulture Extension team has been receiving numerous inquiries about delayed and stunted shoot growth this spring, primarily from western wine production regions of the state. Bud break occurred in mid- to late April for most vineyards in the region, which is considered normal. The region experienced frost events in the last few days of April and beginning of May that affected some vineyards from the Willamette Valley down to the Umpqua, Rogue, Applegate and Illinois Valleys of southern Oregon. Questions started coming in during May as growers began reporting delayed bud break and lagging shoot growth across western Oregon. Many of these growers did not report any frost damage from the late April frost events.
Symptoms reported by growers included the following:
- Delayed and sporadic bud break in cane pruned vines, with greater delay in growth at mid and distal cane positions.
- Stunted or delayed shoot growth, often noticed in shoots on the mid-cane node positions
- Shoots with only inflorescences and no shoot tips on vines with healthy shoots
- Symptoms were reported for both young and mature vines.
Observations by Region
Umpqua Valley. Steve Renquist, OSU Extension Horticulturist in Douglas County, reported more issues in the northern half of Douglas County, primarily in higher elevation sites and hilltop areas of vineyards. Affected vineyards had uneven vine growth and some vines not breaking bud at all, primarily for Spanish and Rhone cultivars, many of which break bud earlier than cool climate cultivars. Canes had green cambium but buds were not pushing, and healthy shoot growth was primarily coming from suckers at the head and trunks of vines. He attributed the symptoms to cold damage caused by prolonged cold weather for two weeks in February combined with persistent snow cover. This time period was also characterized windy, cold nighttime conditions that may have led to further damage.
Southern Oregon. Alex Levin, Assistant Professor at the OSU Southern Oregon Research and Extension Center in Central Point, reported strange early season growth that was suspected to be due to the post-bud break frost events. However, the symptoms differed from typical frost damage and was not found only in low lying areas of vineyards. The problem was most prominent in Pinot noir compared to other cultivars in the region. Growers reported delayed bud break, stunted shoot growth, shoots with no shoot tips, buds that grew flower clusters with no shoot or minimal shoot (Figure 2), and leaf cupping with blackened/necrotic lesions of the leaf blade-petiole juncture (Figure 3). However, by late May, vine growth improved with warmer temperatures and lateral shoot push. It is unclear what caused these symptoms, but they align with those associated with Pinot Leaf Curl, a physiological disorder reported in California.



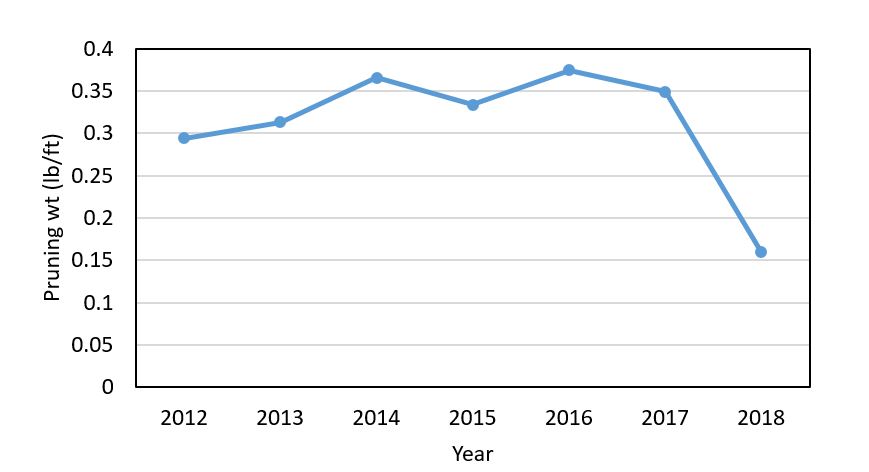
Willamette Valley. There were fewer reports of delayed bud break and stunting in the Willamette Valley than in the southern reaches of the state. However, similar symptoms were reported. In some cases, vineyards with delayed growth were linked to frost damage, vine nutrient stress, or trunk disease. In several cases, the cause of the problem was likely related to a compounding effect of vine stress over several seasons. The dry 2018 growing season took a toll on vine growth that may be manifesting symptoms this season. In examining data over multiple sites and multiple seasons in one of my research projects, I found that pruning weights were lower in 2018 than in the past six seasons (Figure 4). The pruning weights ranged from 0.07 to 0.16 lb/ft across the vineyards, which is lower than the 0.2-0.4 lb/ft that is considered optimum range for plant vigor. Most people reported having full canopies during 2018 that required less hedging; this may suggest fewer carbohydrates were stored in reserves by the post-harvest period.
A lack of reserve carbohydrates and nutrients may lead to reduced shoot growth in spring, as the vine is relying on stored resources for growth after bud break. If a vine is compromised in some way (e.g. trunk disease, crown gall, water stress, etc.) low carbohydrate reserves can further exacerbate issues with early season growth. It is possible that other areas of the state experienced similar declines in grapevine growth in 2018 that may play a role in what is being manifested as 2019 erratic spring growth. Exploring your own historical pruning weight data will help determine if you see a similar trend for 2018.
Weather considerations
Weather also needs to be considered as a factor affecting growth this spring. The 2018-2019 winter was mild until February which brought colder temperatures to most of the region. Mean daily temperatures for the month of February ranged from 36 to 38°F across western Oregon which are 5-6°F lower than the long-term averages for February. The rest of the dormant period (Nov-Mar) differed from the long-term averages by ~1-2°F. Despite being cooler in February, the minimum daily temperatures observed in February in western Oregon (29-33°F) were not within the range known to cause damage to dormant grapevines. However, the temperatures may have caused some tissue damage for vines in ecodormancy, the state at which vine tissues begin to deacclimate for spring. Grapevine tissues (phloem, buds, and xylem) become more sensitive to cold temperatures in the deacclimation phase and can be damaged at increasing temperatures (Ferguson et al. 2011). Fluctuating daily temperatures lead to changes in the vine’s cold hardiness (e.g. low temperature tolerance). The warmer conditions in January may have led to vine deacclimation, leaving tissues more sensitive to temperatures in February. Cool climate cultivars have quicker deacclimation and earlier bud break (Ferguson et al 2014) that may leave them more vulnerable to damage late winter than other cultivars.
Research conducted at OSU described the chilling requirements for Pinot noir to transition to different stages of dormancy and the temperature thresholds that would damage buds at the quiescent stage through to early growth stages. The results showed that 50% of the buds were damaged (LD50) at 6.8 °F, 25.8°F, and 28.0°F for the quiescent, bud swell, and bud burst stages, respectively (Gardea 1988). Another study showed a threshold of 300 cumulative chilling hours to reach ecodormancy in Pinot noir (Gardea 1992). The more chilling that was received during dormancy led to more even bud break in that study. There is also the potential that low temperatures in winter and early spring to affect cell growth and development without causing death of the bud or vascular tissues. Cool winter and spring temperatures can influence vine growth and lead to slowed shoot development, deformed leaves and other physiological disorders that are not well described.
Although temperature data that is available from regional weather stations, such as AgriMet, do not indicate concerning temperatures, check temperature data from on-vineyard weather stations, if available. Fluctuating warm and cold temperatures in late winter, particularly in the January to March period, may explain one potential factor related to irregular growth you may be seeing in your vineyards. Be sure to check the cold temperatures for late April and early May, as post-bud break frost events occurred across western Oregon. These frost events may have also led to some tissue death or irregular growth.
Summary
There are many factors that can lead to delayed bud break and stunted vine growth in spring, including trunk disease, tissue damage due to frost or cold temperature events, nutrient deficiency, vole damage, rust or bud mites, and herbicide damage. However, the delayed growth and stunting reported in vineyards this spring seems to be somewhat consistent across the western region of the state, suggesting an abiotic factor. The weather conditions during late winter and early spring may have played a role in addition to underlying factors that influence vine health and productivity. To learn more about causes of stunting and delayed growth or about cold hardiness of grapevine tissues, be sure to explore the online resources below.
Further Reading
- Recognize the Symptoms and Causes of Stunted Growth in Vineyards, OSU Extension publication and app
- Grapevine Cold Hardiness Monitoring, WSU
- WSU Grapevine Cold Hardiness Model
- Pinot Leaf Curl, UC Cooperative Extension
Literature Cited
Ferguson JC, Tarara JM, Mills LJ, Grove GG, Keller M. 2011. Dynamic thermal time model of cold hardiness for dormant grapevine buds. Ann Bot 107 (3) 389-396.
Ferguson JC, Moyer MM, Mills LJ, Hoogenboom G, Keller M. 2014. Modeling dormant bud cold hardiness and bud break in twenty-three Vitis genotypes reveals variation by region of origin. Am J Enol Vitic 65: 59-71.
Gardea AA. 1988. Freeze damage of Pinot noir (Vitis vinifera L.) as affected by bud development, INA-bacteria, and a bacterial inhibitor. MS Thesis. Oregon State University.
Gardea AA. 1992. Water partitioning and respiration activity of dormant grape buds. PhD Thesis. Oregon State University.







